before
&
after

before
& after
Clinical results with STRIANIX-P® Cream
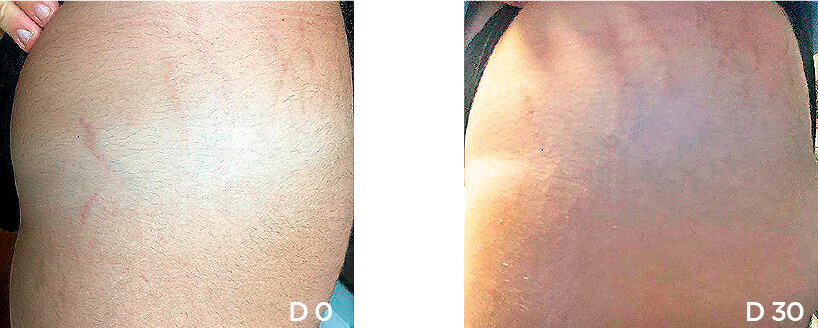
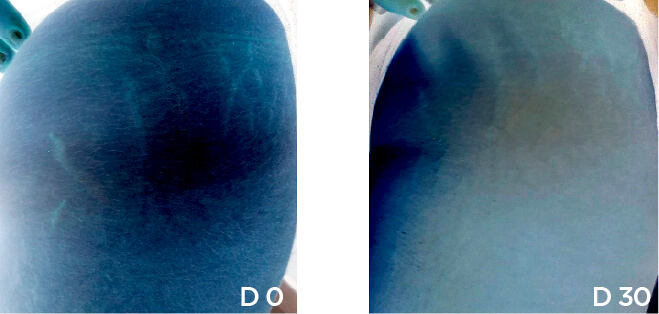
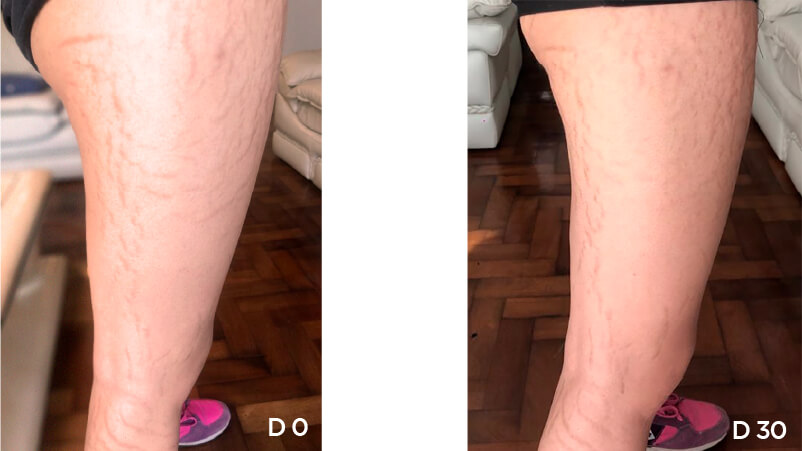
We present our Medical study(*) of patients after 30 days stretch marks treatment with STRIANIX-P® Cream. See here the results of treatment in prevention and management of pink stretch marks.
(*) Clinical trials made by Dra. Graciela Sabbaga (Córdoba, Argentina), Surgeon, Doctor of Medicine and Surgery, Associate Professor of Surgery at the National University of Córdoba, Specialist in general surgery, gynecology and exfoliative cytology.
In case, you still have any questions, ask your doctor or your pharmacist.

before & after

please access the session indicated for you:

Clinical
results

Clinical
results
Evaluation of efficacy and tolerance of STRIANIX-P® in the treatment of striae rubrae (SR)
Study aim: to evaluate the efficacy and tolerance of STRIANIX-P® Cream in the treatment of striae rubrae.
Investigator: Dr Graciela Sabbaga MD, Gynaecologist.
Number of patients: 30.
Duration of study: 90 days.
Posology: STRIANIX-P® Cream 2xday.
Main criterion of evaluation: surface analysis of the lesions of striae rubrae on digital photographs taken at D0, D30, D60 and D90.
© LSI SILDERMA 2022. ALL RIGHTS RESERVED.
Results and conclusion
Demographic data of study
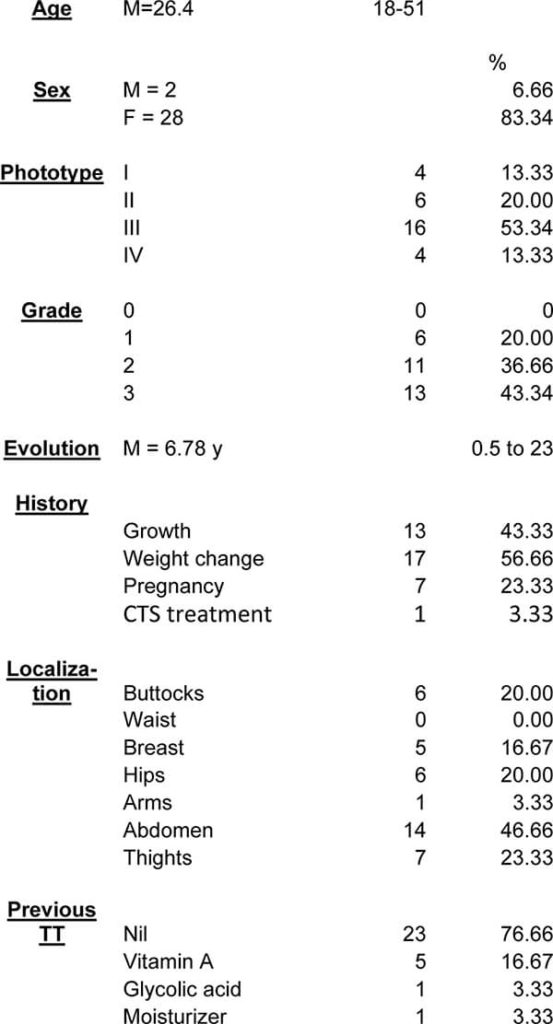
Of 30 enrolled voluntaries, 29 terminated the trial. The patient leaving the study did it for personal reasons; This study was performed during an outbreak of Covid19 and this patient was reluctant to attend a medical office during this period.
Main criterion
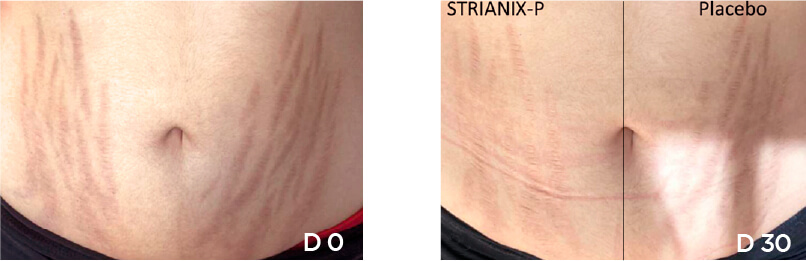
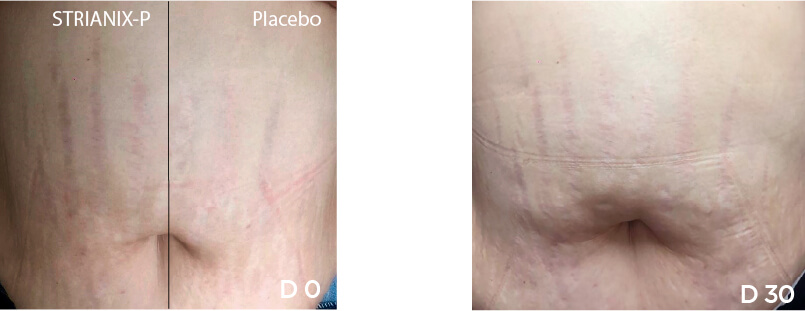
In this study, the main criterion was the reduction of surface of the lesions at D30, D60 and D90 compared with baseline. This reduction was compared to the evolution of the lesions where placebo cream was applied. The results obtained at D30, D60 and D90 with the product under study and with the placebo.
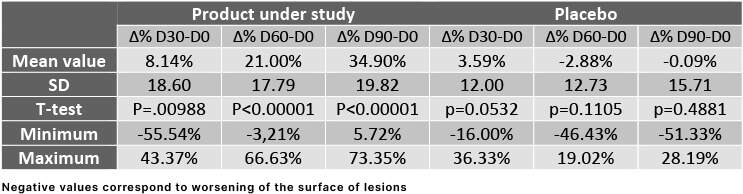
Reduction of the surface of lesions along time
Secondary criterion
The grade of the lesions of striae rubrae was rated between 0 and 3 at each visit V0, V1, V2 y V3.
The scoring is as follows:
0 = No striae
1 = Few thin striae
2 = Many thin striae and few thick striae
3 = Many thick striae
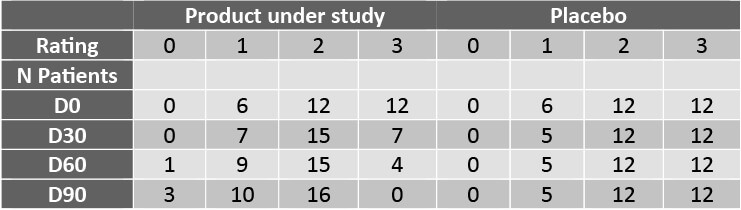
Improvement along the study
Improvement
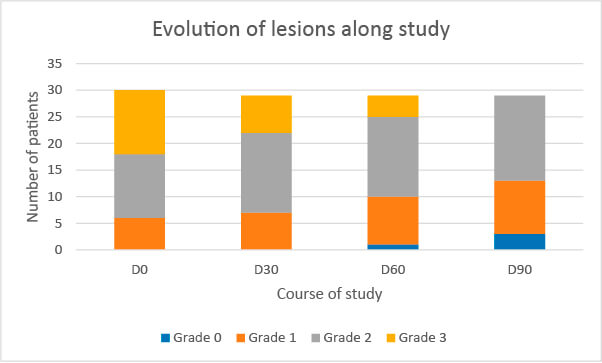
Improvement with the product under study along the trial, compared to D0
Tolerance
Tolerance was quoted as excellent in all the patients at the end of the study, as well with the product under study as with placebo.
No adverse event was reported in any patient.
Conclusions
The product under study showed to be successful in significantly reducing the surface of lesions in patients with striae rubrae, compared to placebo.
Further, the tolerance was quoted as excellent in the absence of any side effect.
Clinical results – Striae Rubrae (SR) Summary
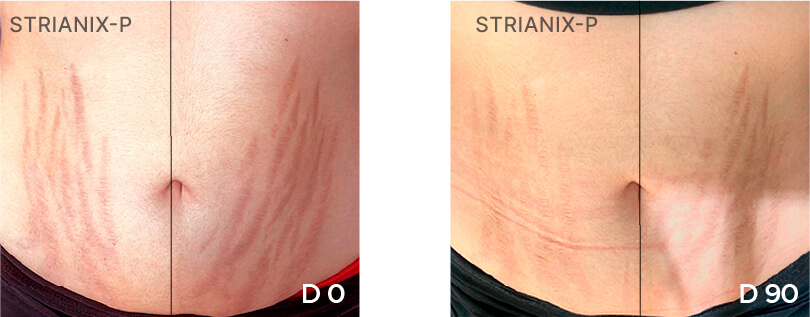
Patient Case 03 – 90 days of treatment with STRIANIX-P® Cream.


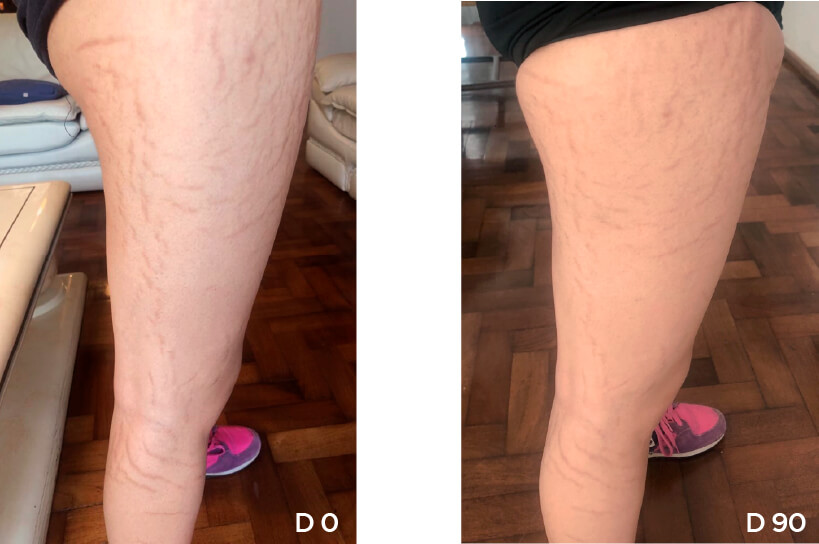
Patient Case 04 – 90 days of treatment with STRIANIX-P® Cream.
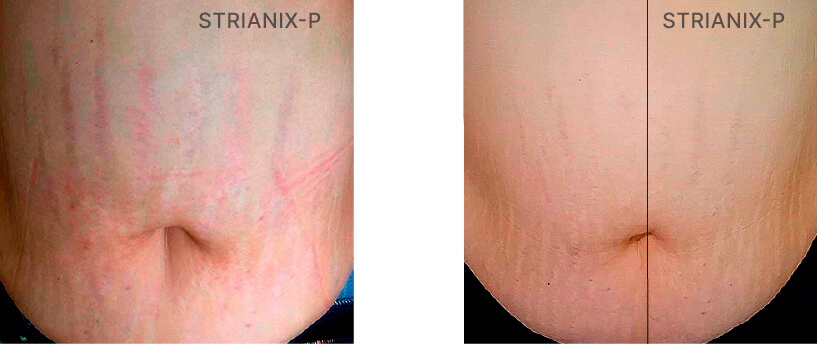
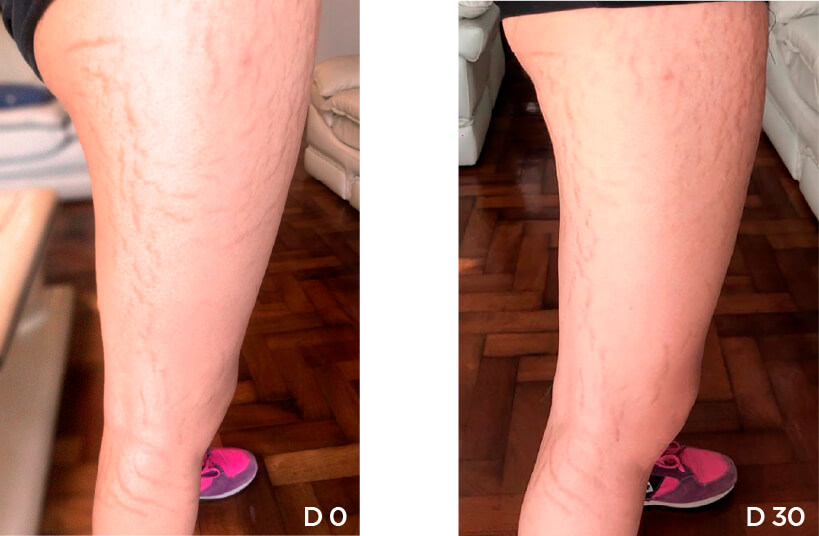
Patient Case 10 – 30 days of treatment with STRIANIX-P® Cream.
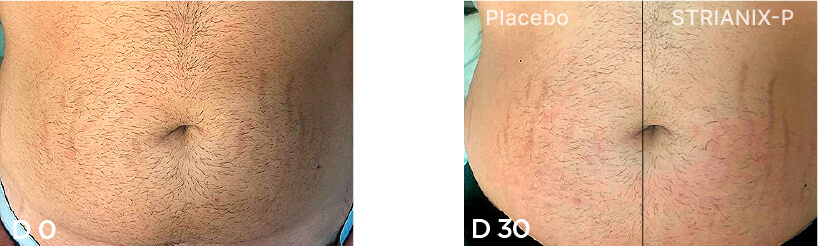
Patient Case 11 – 90 days of treatment with STRIANIX-P® Cream.
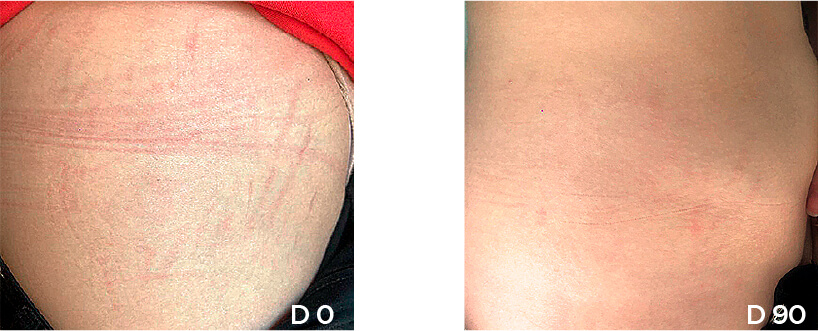
Patient Case 12 – 30 days of treatment with STRIANIX-P® Cream.